Conjugated estrogens (CE) and Bazedoxifene (BZA) combination is used to alleviate menopause-associated symptoms in women. CE+BZA undergo first-pass-metabolism in the liver and deconjugation by gut microbiome via β-glucuronidase (GUS) enzyme inside the distal gut. To date, the impact of long-term exposure to CE+BZA on the gut microbiome or GUS activity has not been examined. Our study using an ovariectomized mouse model showed that CE+BZA administration did not affect the overall cecal or fecal microbiome community except that it decreased the abundance of Akkermansia, which was identified as a fecal biomarker correlated with weight gain. The fecal GUS activity was reduced significantly and was positively correlated with the abundance of Lactobacillaceae in the fecal microbiome. We further confirmed in Escherichia coli K12 and Lactobacillus gasseri ADH that Tamoxifen-, 4-hydroxy-Tamoxifen- and Estradiol-Glucuronides competed for GUS activity. Our study for the first time demonstrated that long-term estrogen supplementation directly modulated gut microbial GUS activity. Our findings implicate that long-term estrogen supplementation impacts composition of gut microbiota and microbial activity, which affects estrogen metabolism in the gut. Thus, it is possible to manipulate such activity to improve the efficacy and safety of long-term administered estrogens for postmenopausal women or breast cancer patients. PMID: 29802368
Category Archives: Metabolism
Bazedoxifene and conjugated estrogen combination maintains metabolic homeostasis and benefits liver health
The bazedoxifene and conjugated estrogens (CE+BZA) combination has been shown to prevent visceral adiposity and weight gain after ovariectomy. However, its impact on the liver transcriptomes associated with prevention of hepatosteatosis is yet to be determined. In the present study, we use liver transcriptomics and plasma metabolomics analysis to characterize the effects of various estrogens on liver. The CE+BZA combination was very effective at preventing ovariectomy-induced weight gain in mice fed a high-fat diet (HFD). In CE+BZA treated animals, liver weight and hepatic lipid deposition were significantly lower than in Vehicle (Veh) treated animals. Additionally, CE+BZA induced unique liver transcriptome and plasma metabolome profiles compared to estradiol, conjugated estrogens alone, and bazedoxifene alone. Blood plasma metabolite analysis identified several metabolites similar to and distinct from other estrogen treatments. Integrated pathway analysis showed that gene networks that were associated with inflammation, reactive oxygen species pathway and lipid metabolism and their relevant metabolites were regulated significantly by CE+BZA treatment. Thus, long-term CE+BZA treatment modulated hepatic metabolic gene networks and their associated metabolites and improves hepatic health without stimulating the uterus. PMID:29267318
Estrogens and female liver health
Due to declining estrogen levels during menopause, NAFLD prevalence is higher in postmenopausal women compared to in premenopausal women or in men. Postmenopausal women are more susceptible to weight gain, fat redistribution and dyslipidemia, all major hallmarks of metabolic syndrome associated with increased NAFLD risk. Gut microbiota plays important roles in development of gastrointestinal tract, metabolism and immunity. Host-microbe interactions allows regulation of a wide range of pathways that affect healthy and diseased physiology. Recent advances in – omics technologies, such as microbiome, transcriptome and metabolome analysis, provided evidence that estrogens and intestinal microbiota (IM) can collectively influence obesity, inflammatory disease, diabetes, and cancers. By understanding underlying mechanisms of estrogens and microbiota crosstalk, we might design dietary and pharmacological interventions to alleviate the metabolic syndrome and NAFLD. PMID:29100781
Non-Nuclear Estrogen Receptor Activation Improves Hepatic Steatosis in Female Mice.
Estrogens have the potential to afford atheroprotection, to prevent excess adiposity and its metabolic complications including insulin resistance, and to lessen hepatic steatosis. Cellular responses to estrogens occur through gene regulation by nuclear estrogen receptors (ER), and through signal initiation by plasma membrane-associated ER. Leveraging the potentially favorable cardiometabolic actions of estrogens has been challenging because their reproductive tract and cancer-promoting effects adversely impact the risk-benefit ratio of the therapy. In prior works we discovered that an estrogen dendrimer conjugate (EDC) comprised of ethinyl-estradiol (E2) molecules linked to a poly(amido)amine dendrimer selectively activates non-nuclear ER, and in mice EDC does not invoke a uterotrophic response or support ER-positive breast cancer growth. In the present investigation, we employed EDC to determine how selective non-nuclear ER activation impacts atherosclerosis, adiposity, glucose homeostasis and hepatic steatosis in female mice. In contrast to E2, EDC did not blunt atherosclerosis in hypercholesterolemic apoE-/- mice. Also in contrast to E2, EDC did not prevent the increase in adiposity caused by western diet feeding in wild-type mice, and it did not affect western diet-induced glucose intolerance. However, E2 and EDC had comparable favorable effect on diet-induced hepatic steatosis, and this was related to downregulation of fatty acid and triglyceride synthesis genes in the liver. Predictably only E2 caused a uterotrophic response. Thus, although non-nuclear ER activation does not prevent atherosclerosis or diet-induced obesity or glucose intolerance, it may provide a potential new strategy to combat hepatic steatosis without impacting the female reproductive tract or increasing cancer risk. Link
Estrogen and Microbiota Crosstalk: Should We Pay Attention?
Recent advances have suggested that steroid hormones such as estrogens, and gut microbiota might synergize to influence obesity, diabetes, and cancer. We discuss recent knowledge of the interactions between estrogens and gut microbiota, and new insights that might offer new approaches to influence this crosstalk and improve metabolic outcomes. Link
Design of pathway preferential estrogens that provide beneficial metabolic and vascular effects without stimulating reproductive tissues
There is great medical need for estrogens with favorable pharmacological profiles that support desirable activities for menopausal women, such as metabolic and vascular protection, but that lack stimulatory activities on the breast and uterus. We report the development of structurally novel estrogens that preferentially activate a subset of estrogen receptor (ER) signaling pathways and result in favorable target tissue–selective activity. Through a process of structural alteration of estrogenic ligands that was designed to preserve their essential chemical and physical features but greatly reduced their binding affinity for ERs, we obtained “pathway preferential estrogens” (PaPEs), which interacted with ERs to activate the extranuclear-initiated signaling pathway preferentially over the nuclear-initiated pathway. PaPEs elicited a pattern of gene regulation and cellular and biological processes that did not stimulate reproductive and mammary tissues or breast cancer cells. However, in ovariectomized mice, PaPEs triggered beneficial responses both in metabolic tissues (adipose tissue and liver) that reduced body weight gain and fat accumulation and in the vasculature that accelerated repair of endothelial damage. This process of designed ligand structure alteration represents a novel approach to develop ligands that shift the balance in ER-mediated extranuclear and nuclear pathways to obtain tissue-selective, non-nuclear PaPEs, which may be beneficial for postmenopausal hormone replacement. The approach may also have broad applicability for other members of the nuclear hormone receptor superfamily. PMID: 27221711
Systems Biology of Metabolic Regulation by Estrogen Receptor Signaling in Breast Cancer
With the advent of the -omics approaches our understanding of the chronic diseases like cancer and metabolic syndrome has improved.
However, effective mining of the information in the large-scale datasets that are obtained from gene expression microarrays, deep sequencing experiments or metabolic profiling is essential to uncover and then effectively target the critical regulators of diseased cell phenotypes. Estrogen Receptor α (ERα) is one of the master transcription factors regulating the gene programs that are important for estrogen responsive breast cancers. In order to understand to role of ERα signaling in breast cancer metabolism we utilized transcriptomic, cistromic and metabolomic data from MCF-7 cells treated with estradiol. In this report we described generation of samples for RNA-Seq, ChIP-Seq and metabolomics experiments and the integrative computational analysis of the obtained data. This approach is useful in delineating novel molecular mechanisms and gene regulatory circuits that are regulated by a particular transcription factor which impacts metabolism of normal or diseased cells. Zhao, Y.C., Madak Erdogan, Z. Systems Biology of Metabolic Regulation by Estrogen Receptor Signaling in Breast Cancer. J. Vis. Exp. (109), e53832, doi:10.3791/53832 (2016).
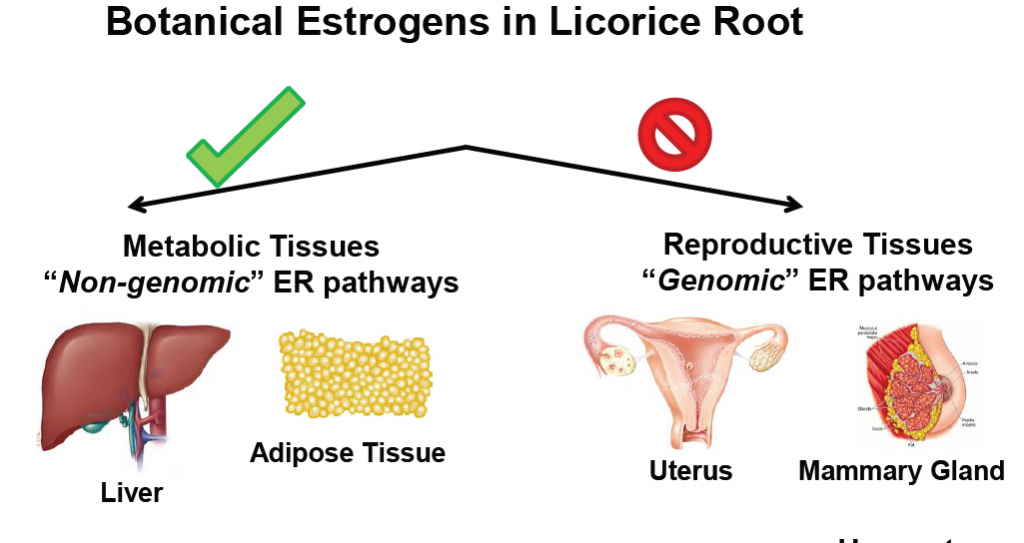
Dietary licorice root supplementation reduces diet-induced weight gain, lipid deposition, and hepatic steatosis in ovariectomized mice without stimulating reproductive tissues and mammary gland
Integrative Genomics of Gene and Metabolic Regulation by Estrogen Receptors α and β and Coregulators, Madak-Erdogan Z, Charn T.H, Jiang Y, Liu E.T., Katzenellenbogen J.A., Katzenellenbogen B.S.Molecular Systems Biology 2013 Jun 18;9:676,
The closely related transcription factors (TFs), estrogen receptors ERα and ERβ, regulate divergent gene expression programs and proliferative outcomes in breast cancer. Utilizing breast cancer cells with ERα, ERβ, or both receptors as a model system to define the basis for differing response specification by related TFs, we show that these TFs and their key coregulators, SRC3 and RIP140, generate overlapping as well as unique chromatin-binding and transcription-regulating modules. Cistrome and transcriptome analyses and the use of clustering algorithms delineated 11 clusters representing different chromatin-bound receptor and coregulator assemblies that could be functionally associated through enrichment analysis with distinct patterns of gene regulation and preferential coregulator usage, RIP140 with ERβ and SRC3 with ERα. The receptors modified each other’s transcriptional effect, and ERβ countered the proliferative drive of ERα through several novel mechanisms associated with specific binding-site clusters. Our findings delineate distinct TF-coregulator assemblies that function as control nodes, specifying precise patterns of gene regulation, proliferation, and metabolism, as exemplified by two of the most important nuclear hormone receptors in human breast cancer. PMID: 23774759
Non-nuclear Estrogen Receptor α Signaling Promotes Cardiovascular Protection but not Uterine or Breast Cancer Growth in Mice,Chambliss K.L, Wu Q., Oltmann S, Umetani M, Korach K.L., Thomas G.D., Mineo C., Yuhanna I.S., Kim S.H., Madak-Erdogan Z., Maggi A., Dineen S.P., Roland C.L., Brekken R.A., Katzenellenbogen J.A., Katzenellenbogen B.S., Shaul P.W., Journal of Clinical Investigation, 2010,120(7):2319:2330
Steroid hormone receptors function classically in the nucleus as transcription factors. However, recent data indicate that there are also non-nuclear subpopulations of steroid hormone receptors, including estrogen receptors (ERs), that mediate membrane-initiated signaling of unclear basis and significance. Here we have shown that an estrogen-dendrimer conjugate (EDC) that is excluded from the nucleus stimulates endothelial cell proliferation and migration via ERalpha, direct ERalpha-Galphai interaction, and endothelial NOS (eNOS) activation. Analysis of mice carrying an estrogen response element luciferase reporter, ER-regulated genes in the mouse uterus, and eNOS enzyme activation further indicated that EDC specifically targets non-nuclear processes in vivo. In mice, estradiol and EDC equally stimulated carotid artery reendothelialization in an ERalpha- and G protein-dependent manner, and both agents attenuated the development of neointimal hyperplasia following endothelial injury. In contrast, endometrial carcinoma cell growth in vitro and uterine enlargement and MCF-7 cell breast cancer xenograft growth in vivo were stimulated by estradiol but not EDC. Thus, EDC is a non-nuclear selective ER modulator (SERM) in vivo, and in mice, non-nuclear ER signaling promotes cardiovascular protection. These processes potentially could be harnessed to provide vascular benefit without increasing the risk of uterine or breast cancer. PMID:20577047